"Innovative technology in a handy device"

Box
Gerät
„Medical technology at its best“
Great technology in a handy device
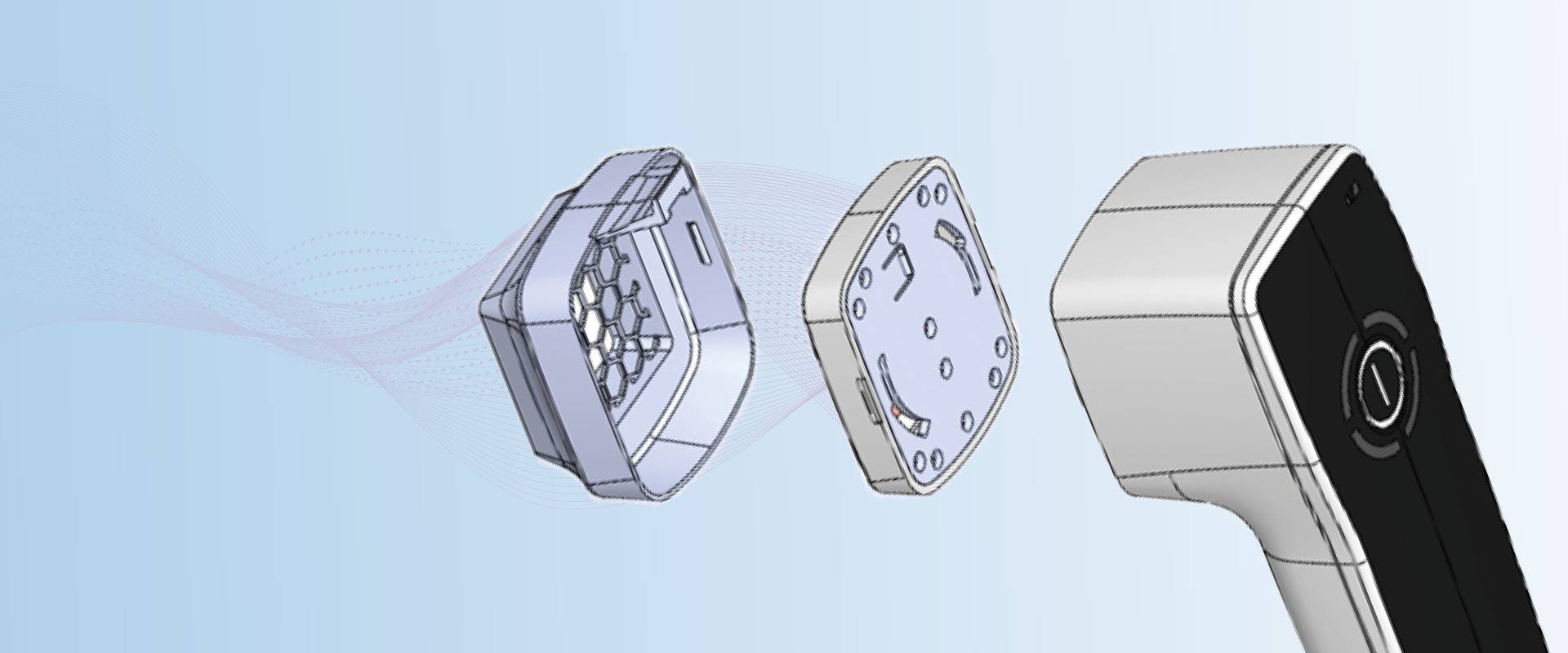
The plasma care® product line is handily designed for mobile use by doctors and nurses. The device is functional, user-friendly and at the same time aesthetically designed. The spacer – also known as spacer – guarantees hygienic use.

1. The cover with touch button to turn the device on and off, the housing with ergonomic design includes all the electrics and control of the device.

2. The plasma source – is the heart of plasma care®, where the plasma is generated by microdejections. It is equipped with wafer-thin ceramic plates and sensitive electrical contacts.

3. The sterile disposable spacer is used per treatment. It is mounted on the plasma source and guarantees the hygienic use of the device – even in busy everyday practice. A new spacer must be used for each treatment, which is ensured by an RFID unit (radio-frequency identification); it turns off the device when a spacer is used repeatedly and displays this accordingly in the user interface..

4. Docking station: The batteries of the plasma care® are charged wirelessly through the docking station to avoid the ingress of liquid and dirt into the housing. Inductive charging ensures that the user and the patient cannot have contact with live components. Once charged, the battery lasts for about 120 applications

5. The power supply: The power supply is connected to the docking station

All housing parts are made of medical plastics. The surfaces are made of biocompatible plastics. The controls and LED lights are integrated into the housing in such a way that there are no openings – the advantages: dirt cannot be placed, deposits are avoided, easy cleaning with wipe disinfection is made possible.
Plasma technology – it brings lightning to room temperature
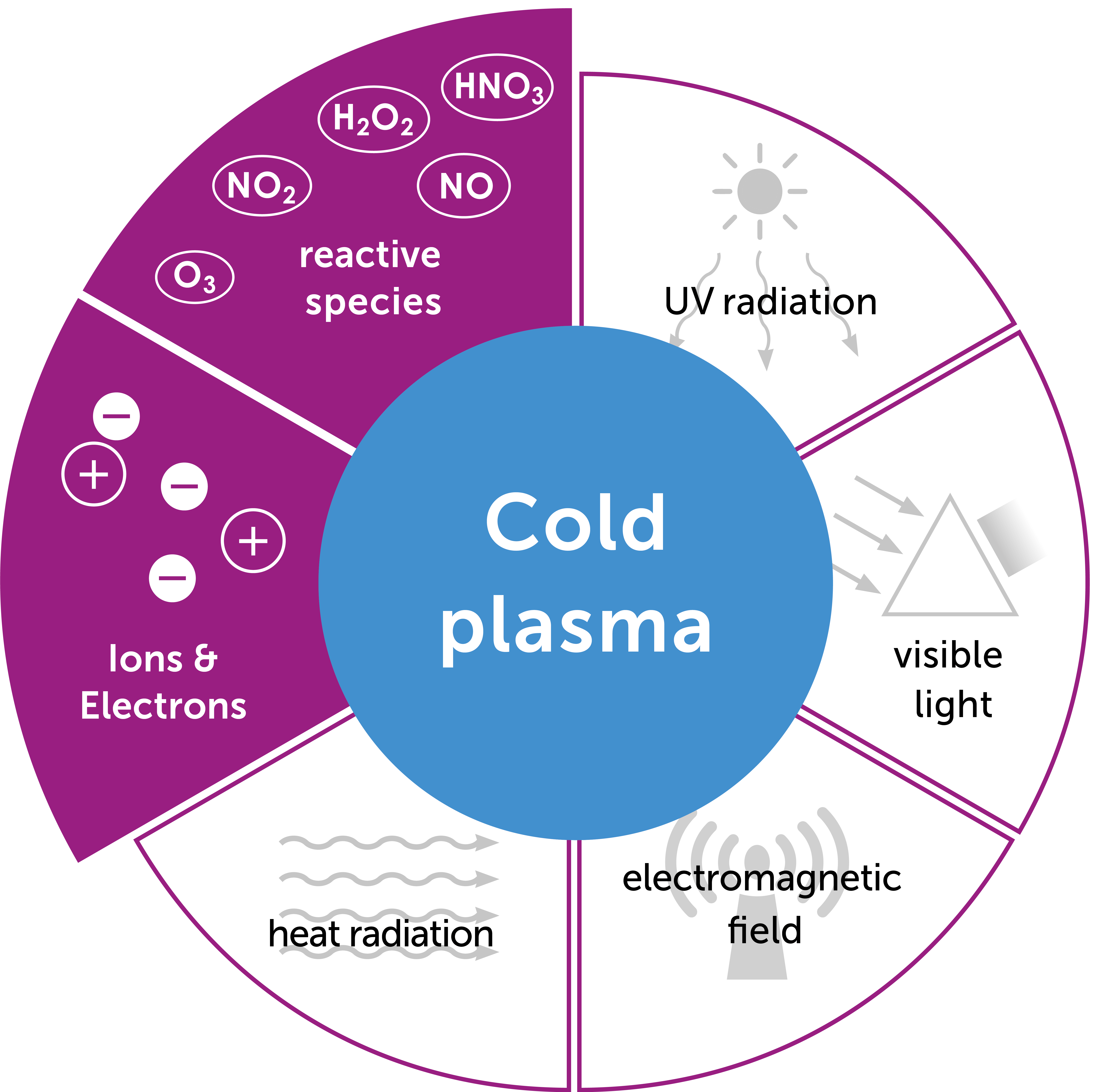
Plasma is an ionized gas, i.e. it contains charged particles. It is formed from a gas to which energy is supplied, usually in the form of heat, and is often referred to as the fourth state of matter. Natural examples of plasmas are the sun or lightning on Earth.
In addition to its medical application, cold plasma has many other positive effects. You can find out more about this at terraplasma.
The photo shows a picture of a plasma source. Fig. 1: Microdischarge in the electrode 2: Plasma source in the darkened environment in which the characteristic purple glow of the plasma becomes visible..
Cold atmospheric plasmas (CAP) are partially ionized gases, i.e. only one particle from 1∙109 is ionized. The advantage of these cold atmospheric plasmas is that they can persist at room temperature and atmospheric pressure. Plasma care® uses patented thin-film technology, a further development of surface micro-discharge technology (SMD). The Plasma Source Unit consists of a high-voltage electrode, a dielectric and the grounded structured electrode. By applying a high voltage of 3.5 kV, micro-discharges with an expansion of a few millimeters occur in the electrode structured in squares. These ionize the gas and thus generate the plasma.
Plasma Design
The composition of a cold atmospheric plasma is not a fixed quantity, but can be influenced, among other things, by the choice of plasma source and its operating parameters. This is referred to as the so-called “plasma design”. In the case of SMD cold plasma technology, which is used in plasma care® for wound treatment, the plasma design was chosen in such a way that a high bactericidal effect is achieved in vitro and tissue damage is avoided
Similar to Shimizu et al. 2017 DOI: 10.1615/PlasmaMed.2017019455), investigations were carried out with the plasma care® plasma source in so-called oxygen and nitrogen mode to determine the optimal operating parameters for maximum effectiveness and safety. With the help of these and extensive further investigations, plasma care® was carefully developed for its intended purpose.
Patents
The plasma care® product group uses the following patents:
Plasma device for the treatment of body surfaces
Device patent plasma care® – a medical device for the treatment of wounds with cold atmospheric plasma
- Owners: terraplasma GmbH und terraplasma medical GmbH
- Priority date: 15.06.2018
- Patent applications: Deutschland: DE 10 2018 209 735.6, PCT/EP2019/065570
Method for testing an electrode arrangement for the generation of a non-thermal plasma and plasma source with such an electrode arrangement, established for carrying out such a method
Procedure for pre-checking the functionality of a plasma source
- Owners: terraplasma GmbH und terraplasma medical GmbH
- Priority date: 15.06.2018
- Patent applications: Deutschland: DE 10 2018 209 730.5, PCT/EP2019/065574
Electrode arrangement and plasma source for the generation of a non-thermal plasma as well as a method for operating a plasma source
Electrode design – based on surface microdischarge technology, which generates a homogeneous plasma with low voltages of 3-4 kV (thin film technology)
- Owners: terraplasma GmbH (Deutschland und Europa) und terraplasma medical GmbH (China und Japan)
- Priority date: 23.07.2015
- Patent applications: Deutschland: 10 2015 213 975.1, Japan: 2018-522866, Europa: 16744694.7
- Patents: USA: US10470285B2 issued at 10.06.2019, China: ZL 201680043155.0 issued at 05.03.2021
Device patent plasma intensive care® – a system for the treatment and prevention of viral, bacterial
Owner: terraplasma medical GmbH
Priority date: 09.07.2020,
Patent application: EP 20 18 5085.6
Patents: Europa, mit Ausweitung für Japan, USA
Exclusive Licenses
The plasma care® product group uses the following exclusive licenses:
Electrode arrangement and plasma source for the generation of a non-thermal plasma as well as a method for operating a plasma source
Electrode design – based on surface microdischarge technology, which generates a homogeneous plasma with low voltages of 3-4 kV (thin film technology)
- Owner: terraplasma GmbH
- Priority date : 23.07.2015
- Patent application: Deutschland: 10 2015 213 975.1, Japan: 2018-522866, Europa: 16744694.7
- Patents: USA: US10470285B2 issued at 10.06.2019, China: ZL 201680043155.0 issued at 05.03.2021
Optimization of bactericidal effect
Device and method for plasma treatment of surfaces, as well as use of a device
Owner: Max-Planck-Gesellschaft
Priority date: 25.05.2011
Patent application: Europe
ContaCt & FAQ
Questions cold plasma therapy
There are many questions about cold plasma therapy: Which bacteria does cold plasma affect? How long does the therapy last …. We have compiled the most frequently asked questions and answers for you here. You are also welcome to contact us directly:

